The micronucleus test according to DIN EN ISO 21427-2 T5 2020-07 /OECD No.487 is an in vitro method for the identification of damages to chromatin and thereby contributes to the recognition of a cancer risk.
Thus far the method has been limited to recording the biomarker “micronucleus”, a chromatin-containing structure that occurs in addition to the main nucleus, in cultures of mammalian cells as a consequence of exposure to a test substance. Micronuclei/small nuclei have so far been detected after fixation and chromatin staining of the exposed cells.
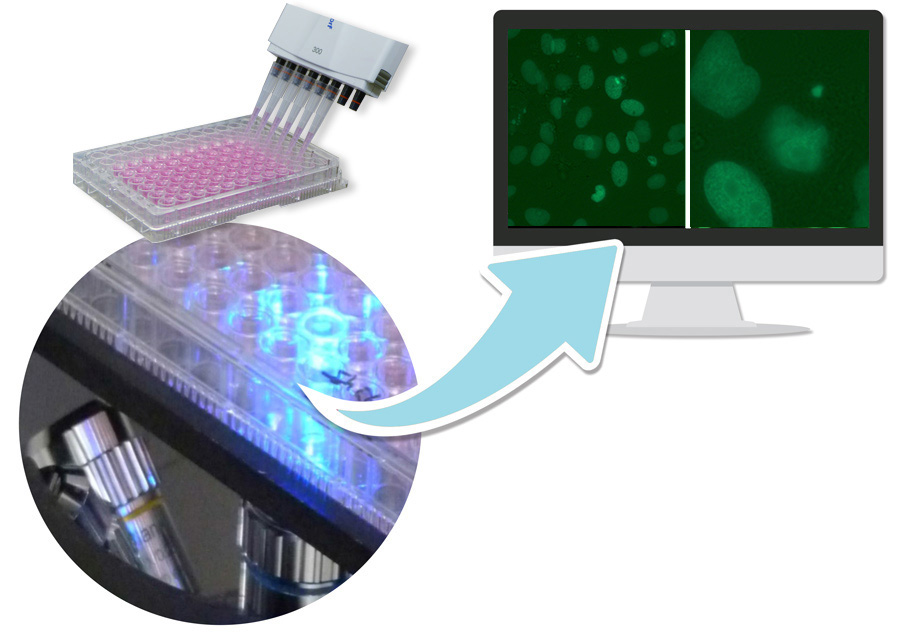
GOBIO has developed an innovative variant of the method as accredited in-house method HM 21427-2, in which micronuclei on living cells can be detected by means of live image analysis. The method is based on a healthy cell line expressing a histone 2B green fluorescence fusion protein. As a result, chromatin structures such as micronuclei are visible in the living cell using fluorescence microscopy. The cells that GOBIO uses for the micronucleus test are neither derived from tumors nor immortalized in any other way. They have a natural control of the cell cycle and are metabolically competent.
They are capable of natural cell death. Due to these properties, the biomarker “micronucleus” can also be used to test the transformation of a previously healthy cell into a cancer cell. The following changes in nuclear morphology occur when a malignant transformation occurs.
Test Procedure
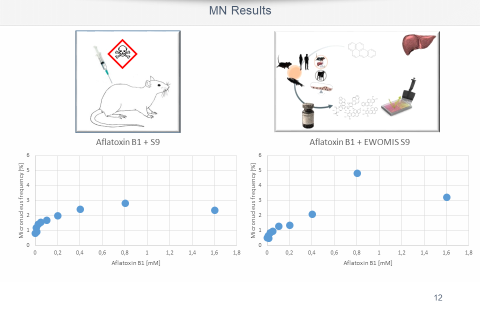
Usually a rangefinder test is carried out first to determine cytotoxicity. The test is performed on 96 well plates. Exposure to pollutants is performed in 4 subcytotoxic concentrations (12 wells per concentration) when the adherent growing cells have reached a confluence of 60 %. Positive and negative controls are included in each experiment. The micronucleus frequency in test approaches is determined using 2 automatically generated images/pro well software. To identify micronuclei, both the fluorescence image and the superposition with the bright field image are analysed.
On customer request the measurement can be performed at different times. This allows the detection of reversible effects, e.g. the reintegration of micronuclei during interphase.
Please leave your contact details if you wish to receive further information. Link to the contact form.
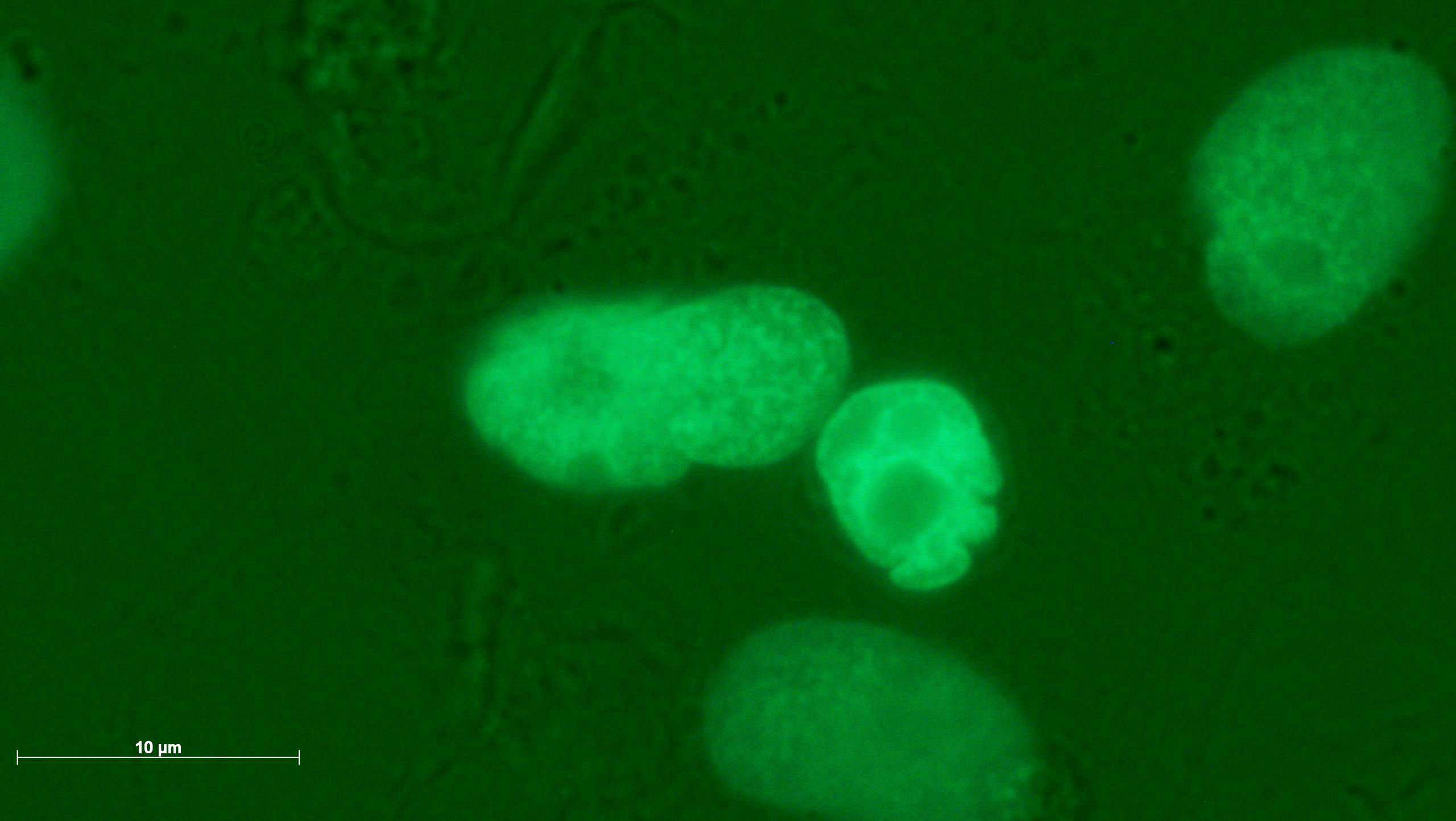
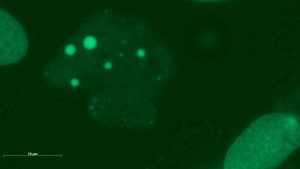
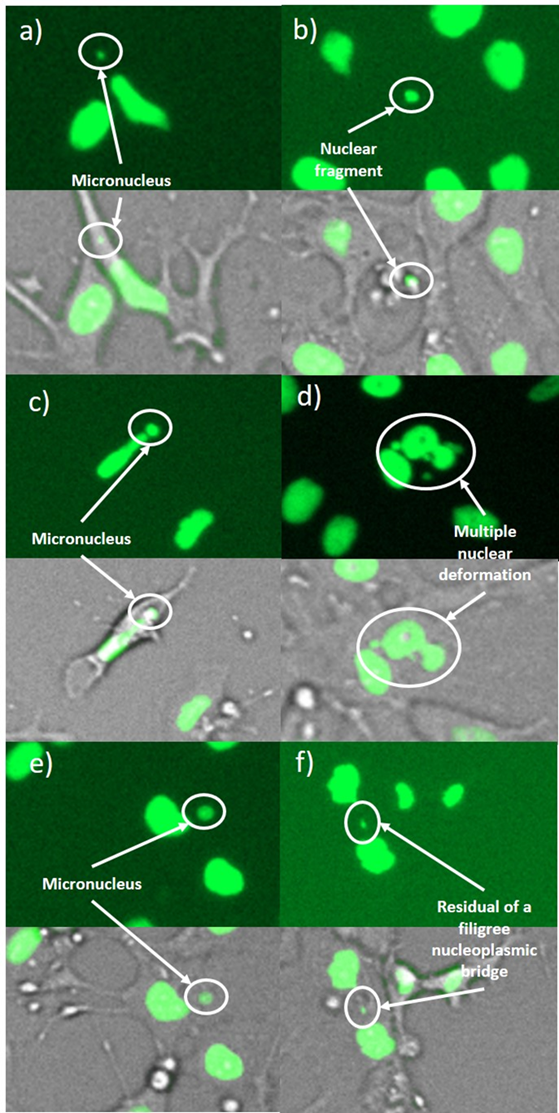
A genotoxic damage pattern comprising (a,b) micronuclei, (c) a large micronucleus, (d) a multiple deformed nucleus, (e) a nuclear fragment outside a cell, and (f) a residue of a filigree nucleoplasmic bridge. The images were taken in 4-NQO exposed cultures (a,b,d), in colchicine exposed cultures (c,e) and in cannabidiol exposed cultures (f).
The benefits of the new, cost-effective micronucleus test
Bastian Niklas Hölzel, Kurt Pfannkuche, Bernhard Allner, Hans Thomas Allner, Jürgen Hescheler, Daniel Derichsweiler, Henner Hollert, Andreas Schiwy, Julia Brendt, Michael Schaffeld, Alexander Froschauer & Petra Stahlschmidt-Allner (2020): Following the adverse outcome pathway from micronucleus to cancer using H2B-eGFP transgenic healthy stem cells. Zur Veröffentlichung.
- Performance characteristics according to DAkkS-tested SOP:
Detection of cytogenetic damage to non-immortalized, healthy cells with functional proliferation control.
- The basic functions of the method are in accordance with OECD Guideline No. 487/ DIN EN ISO 21427-2 T5, i.e. the detection of cytogenetic damage primarily arised during mitosis. Since the culture, unlike synchronously dividing immortalised cells, also includes long living stem cells, damages occurring during interphase are also reliably detected.
- The method is mode of action related. Sub mechanisms are also recorded.
- The test thus goes beyond the performance characteristics of OECD Guideline No. 487/ DIN EN ISO 21427-2 T5
- Due to the multitude of cellular functions that can be observed in the H2B-GFP-KCB cell line, also non-dose-related effects are considered relevant , e.g. interventions in the genetic basis of motility
- Therefore, the criterion of dose dependence is reported, but it is not an exclusion criterion for the assessment of genotoxicity.
- Timely results: 14 days after providing the test substance/test material
- GLP-compliant test report
- Extensive electronic, exposé-suited documentation of the evaluation of the image files
- Cytotoxicity test included at no additional cost
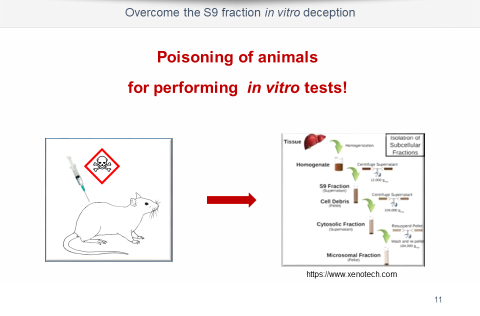
- Animal-free, metabolic activation of the test substance available
- Reversibility can be detected by measuring at different time points.
- Further parameters of the cells’ state of health can be retrieved at low cost, e.g.mitotic index, motility. Downstream analyses possible: cytokinese/interphase duration, apoptosis pycnotisation, gene expression analyses based on a known expressome.